Aznar García M, Canto Mangana J, Martínez de la Plata JE
Hospital Pharmacy Service. Hospital de Poniente. El Ejido. Almería (Spain)
Fecha de recepción: 04/08/2021 – Fecha de aceptación: 31/08/2021
Correspondencia: María Aznar-García – Hospital de Poniente (Hospital Pharmacy Service) – Ctra Almerimar 31 – 04700 El Ejido, Almería (España)
maria.23f.ag@gmail.com
_____
SUMMARY
Non-antiarrhythmic drugs may induce QT-prolongation and increase the risk of arrhythmias. Recent studies have determined that there is a risk of atrial fibrillation (AF) due to QT prolongation. We report a case of FA associated to QT prolongation secondary to a single dose of hydroxychloroquine (HCQ) in an 83-years-old polymedicated patient admitted to our hospital due to SARS-CoV-2 infection. Quetiapine was prescribed as regular medicine after admission and a 5-days oral HCQ regimen was started for COVID-19. Thirty minutes after HCQ loading dose, FA was reported on electrocardiogram (EKG). COVID-19 treatment is leading to use off-label drugs that may generate adverse effects. It should be considered that drugs that induce QT prolongation may be triggers for atrial arrhythmias. There is not any report of sudden onset of increased QT interval with associated arrythmia after a single dose of HCQ, even in a short course treatment.
Key words: Hydroxychloroquine, prolonged QT interval, COVID-19, atrial fibrillation, adverse drug reactions, drug interactions.
Fibrilación auricular y prolongación del QT después de una dosis única de hidroxicloroquina
RESUMEN
Los fármacos no antiarrítmicos pueden inducir la prolongación del intervalo QT y aumentar el riesgo de arritmias. Estudios recientes han determinado que existe riesgo de desarrollar fibrilación auricular (FA) asociada a la prolongación del intervalo QT. Presentamos un caso de FA asociado a prolongación del QT secundario a una dosis única de hidroxicloroquina (HCQ) en una paciente polimedicada de 83 años ingresada en nuestro hospital por infección por SARS-CoV-2. A la paciente se le prescribió quetiapina como parte de su medicamento habitual al ingreso y se inició tratamiento frente a COVID-19 basado en HCQ oral. Treinta minutos tras la dosis de carga de HCQ, se informó FA en el electrocardiograma (ECG). El tratamiento de COVID-19 está llevando al uso de medicamentos no aprobados que pueden generar efectos adversos. Además, debe considerarse que los fármacos que inducen la prolongación del QT pueden desencadenar arritmias auriculares. No se han reportado casos de aparición repentina de aumento del intervalo QT con arritmia asociada después de una dosis única de HCQ.
Palabras clave: Hidroxicloroquina, intervalo QT prolongado, COVID-19, fibrilación auricular, reacciones adversas a medicamentos, interacciones medicamentosas.
____
INTRODUCTION
Non-antiarrhythmic drugs may induce QT-prolongation and increase the risk of arrhythmias and fatal outcome1. QT-interval is a marker of ventricular repolarization and its prolongation is associated with an increased risk of ventricular arrhythmia. Recent studies have established the association between atrial arrythmias and QT prolongation, determining that there is a risk of atrial fibrillation (AF) due to QT prolongation2. According to American Heart Association, is considering QTc prolonged interval when its value is 460 ms or longer in women and 450 ms or longer in men3. Hydroxychloroquine (HCQ), is a drug recently used to treat COVID-19 and know to prolong QTc interval on chronic treatment or poisoning4,5. We report a case of AF associated to QT prolongation secondary to a single dose of HCQ in a polymedicated patient admitted to our hospital due to SARS-CoV-2 infection.
CASE SUMMARY
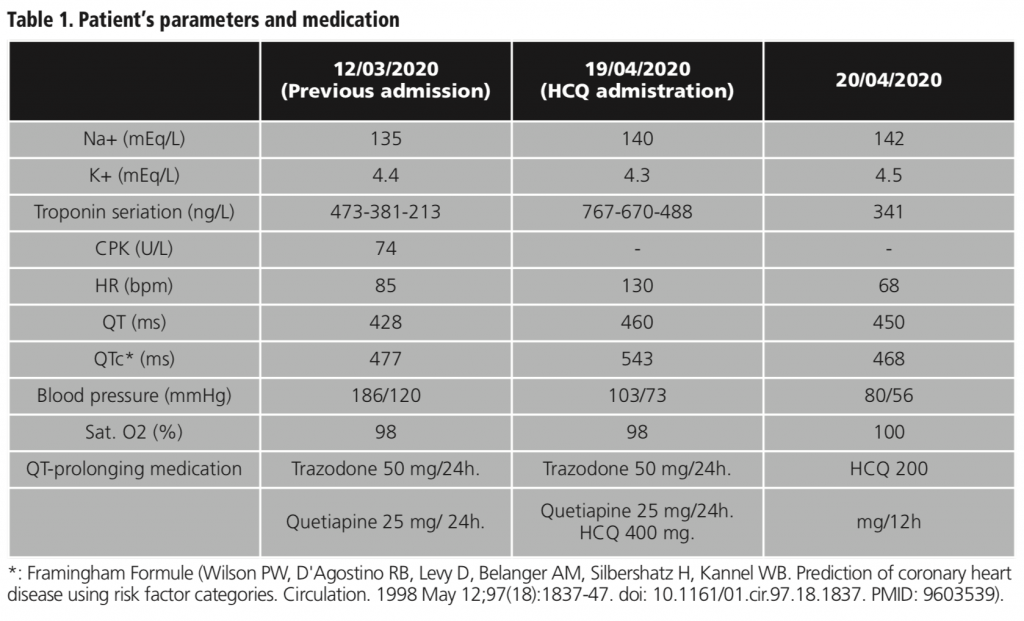
An 83-year-old nursing home resident was brought to Emergency Department in May 2020 due to hypertension, discomfort and diarrhea. Medical past history included left bundle branch block, hypertension, severe dementia and Parkinson’s disease. Atenolol, furosemide, memantine, co-careldopa, trazodone and quetiapine were her regular medicines. She had a previous admission in March 2020 for chest pain secondary to arterial hypertension, EKG and analytic was done (table 1).
On admission, COVID-19 PCR test was done resulting positive. A 5-days oral HCQ regimen was started for COVID-19 based on a loading dose of 400 mg every 12 hours the first day following 200 mg every 12 hours. Further, after admission, quetiapine was prescribed as regular medicine at usual dose (100 mg every 24h).
Thirty minutes after 400 mg HCQ loading dose, the patient had an episode of hypotension and frailty. Electrocardiogram (EKG) was done and AF was reported. The ventricular rate was 130-140 beats per minute and the QTc interval was 543 ms. An increase of 13.83 % on the QTc interval was observed on EKG compared to the previous electrocardiogram in March 2020 (QTc 477ms). The patient was monitorized, troponins and electrolytes were seriated (table 1). Amiodarone perfusion was used to treat the AF. Consequently, quetiapine was stopped due to its potential interaction with HCQ in prolonging the QT interval. The day after quetiapine interruption, a new EKG was performed where it is observed a decrease in QTc of 16.02% (QTc 543 ms to 468 ms). The patient continued her COVID-19 treatment with HCQ without presenting further cardiac events and after her stabilization she was discharged to her residence.
According to Naranjo Scale a score of 7 was obtained indicating a “probable” adverse reaction to HCQ6. Karch and Lasagna’s Algorithm to determine the causal relationship was performed resulting a score of 5 classified as “possible”7. To evaluate the interaction between HCQ-Quetiapine-Trazodone Horn scale was applied, a score of 5 was resulted graded as “probable”8.
Advanced age, female gender, previous heart disease history, electrolyte disorders, and concomitant use of potential torsadogenic medicines are predisposing factors for the QT prolongation and arrythmias risk1.
Our patient had several risk factors and was on quetiapine and trazodone as regular medicines for more than a year ago. Both drugs are classified as “Conditional Risk of TdP” drugs, according to the Arizona Center for Education and Research on Therapeutics (AzCERT). The appearance of arrythmias with these drugs is uncommon and it is usually associated with poisoning or concomitant use of QT-prolonging drugs9.
HCQ has been used as an antimalarial for 70 years, and is now used to treat rheumatoid arthritis and systemic lupus, it have been proposed as a hopeful drug, included in numerous therapeutic protocols against SARS-CoV-2 at the beginning of the pandemic4. According to AzCERT HCQ is classified as “Know Risk of TdP” drug; though TdP has been reported as a HCQ uncommon adverse effect. Normally, HCQ-induced prolonged QT has been showed on chronic treatment or poisoning patients5.
Our patient immediately experienced FA associated to QT prolongation after a single dose of HCQ as a third drug with torsadogenic properties. Due to its pharmacokinetics, HCQ is rapidly absorbed after oral administration and has been observed a lag time before absorption of 0.57 hours10. This may explain a timeline relationship between taking a single dose of HCQ and the onset of FA due to the additive effect with chronic quetiapine and trazodone. So far, there is not any report of sudden onset of increased QT interval with associated arrythmia after a single dose of HCQ, even in a short course treatment.
CONCLUSION
COVID-19 treatment is being a big challenge leading to use off-label drugs that may generate serious adverse effects. Further, due to the relationship between QT prolongation and AF, it should be considered that drugs that induce QT prolongation may be triggers for atrial arrhythmias. The sudden adverse reaction described highlight the need for close monitoring, especially complex patients, polypharmacy or with severe frailty who are going to take HCQ even from the first dose. This case has notified to the corresponding Pharmacovigilance Center.
Conflict of interests: The authors declare that they do not present a conflict of interest.
BIBLIOGRAPHY
1. Cubeddu LX. Drug-induced Inhibition and Trafficking Disruption of ion Channels: Pathogenesis of QT Abnormalities and Drug-induced Fatal Arrhythmias. Curr Cardiol Rev. 2016;12(2):141-54. doi: 10.2174/1573403×12666160301120217. PMID: 26926294; PMCID: PMC4861943.
2. Zhang N, Gong M, Tse G, Zhang Z, Meng L, Yan BP, Zhang L, Wu G, Xia Y, Xin-Yan G, Li G, Liu T. Prolonged corrected QT interval in predicting atrial fibrillation: A systematic review and meta-analysis. Pacing Clin Electrophysiol. 2018 Mar; 41(3):321-327. doi: 10.1111/pace.13292. Epub 2018 Feb 16. PMID: 29380395.
3. Rautaharju PM, Surawicz B, Gettes LS, Bailey JJ, Childers R, Deal BJ, et al. American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; American College of Cardiology Foundation; Heart Rhythm Society. AHA/ACCF/HRS recommendations for the standardization and interpretation of the electrocardiogram: part IV: the ST segment, T and U waves, and the QT interval: a scientific statement from the American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; the American College of Cardiology Foundation; and the Heart Rhythm Society. Endorsed by the International Society for Computerized Electrocardiology. J Am Coll Cardiol. 2009 Mar 17;53(11):982-91. doi: 10.1016/j.jacc. 2008.12.014. PMID: 19281931.
4. RECOVERY Collaborative Group, Horby P, Mafham M, Linsell L, Bell JL, Staplin N, Emberson JR, Wiselka M, Ustianowski A, Elmahi E, Prudon B, Whitehouse T, Felton T, Williams J, Faccenda J, Underwood J, Baillie JK, Chappell LC, Faust SN, Jaki T, Jeffery K, Lim WS, Montgomery A, Rowan K, Tarning J, Watson JA, White NJ, Juszczak E, Haynes R, Landray MJ. Effect of Hydroxychloroquine in Hospitalized Patients with Covid-19. N Engl J Med. 2020 Nov 19;383(21):2030-2040. doi: 10.1056/NEJMoa2022926. Epub 2020 Oct 8. PMID: 33031652; PMCID: PMC7556338.
5. Doyno C, Sobieraj DM, Baker WL. Toxicity of chloroquine and hydroxychloroquine following therapeutic use or overdose. Clin Toxicol (Phila). 2020 Sep 22:1-12. doi: 10.1080/15563650.2020.1817479. Epub ahead of print. PMID: 32960100.
6. Naranjo CA, Busto U, Sellers EM, Sandor P, Ruiz I, Roberts EA, Janecek E, Domecq C, Greenblatt DJ. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981 Aug;30(2):239-45. doi: 10.1038/clpt. 1981.154. PMID: 7249508.
7. Karch FE, Lasagna L. Toward the operational identification of adverse drug reactions. Clin Pharmacol Ther. 1977 Mar;21(3):247-54. doi: 10.1002/cpt 1977213247. PMID: 837643.
8. Horn JR, Hansten PD, Chan LN. Proposal for a new tool to evaluate drug interaction cases. Ann Pharmacother. 2007 Apr;41(4):674-80. doi: 10.1345/aph. 1H423. Epub 2007 Mar 27. PMID: 17389673.
9. Aronow WS, Shamliyan TA. Effects of atypical antipsychotic drugs on QT interval in patients with mental disorders. Ann Transl Med. 2018 Apr;6(8):147. doi: 10.21037/atm.2018.03.17. PMID: 29862236; PMCID: PMC5952011.
10. Tett SE, Cutler DJ, Day RO. Bioavailability of hydroxychloroquine tablets assessed with deconvolution techniques. J Pharm Sci. 1992 Feb;81(2):155-9. doi: 10.1002/jps.2600810211. PMID: 1545355.
_____